
The first and only Non-Invasive Blood-Based RNA Gene Expression diagnostic test that provides guidance for controlled reduction of immunosuppression, by confirming immune quiescence for rejection in post therapy adjustment for Liver Transplant recipients
TruGraf® Liver Confirms Immune Quiescence and Offers Biomarker Guidance for Immunosuppression
Liver transplant recipients feel the burden of immunosuppression.
27%
of LTRs with grafts >1 year die of malignancy or infection 1
12%
experience liver rejection in the first year 2
20%
experience chronic renal failure in the first 5 years of graft life 1
40%
develop post-transplant diabetes mellitus 1
45%
develop dyslipidemia 1
How do you ensure patients receive optimal immunosuppressant therapy?
A New Way to Optimize Immunosuppression
Introducing TruGraf® Liver, the first and only Non-Invasive Blood-Based RNA Gene Expression diagnostic test that provides guidance for controlled reduction of immunosuppression, by confirming immune quiescence for rejection in post therapy adjustment for Liver Transplant recipients
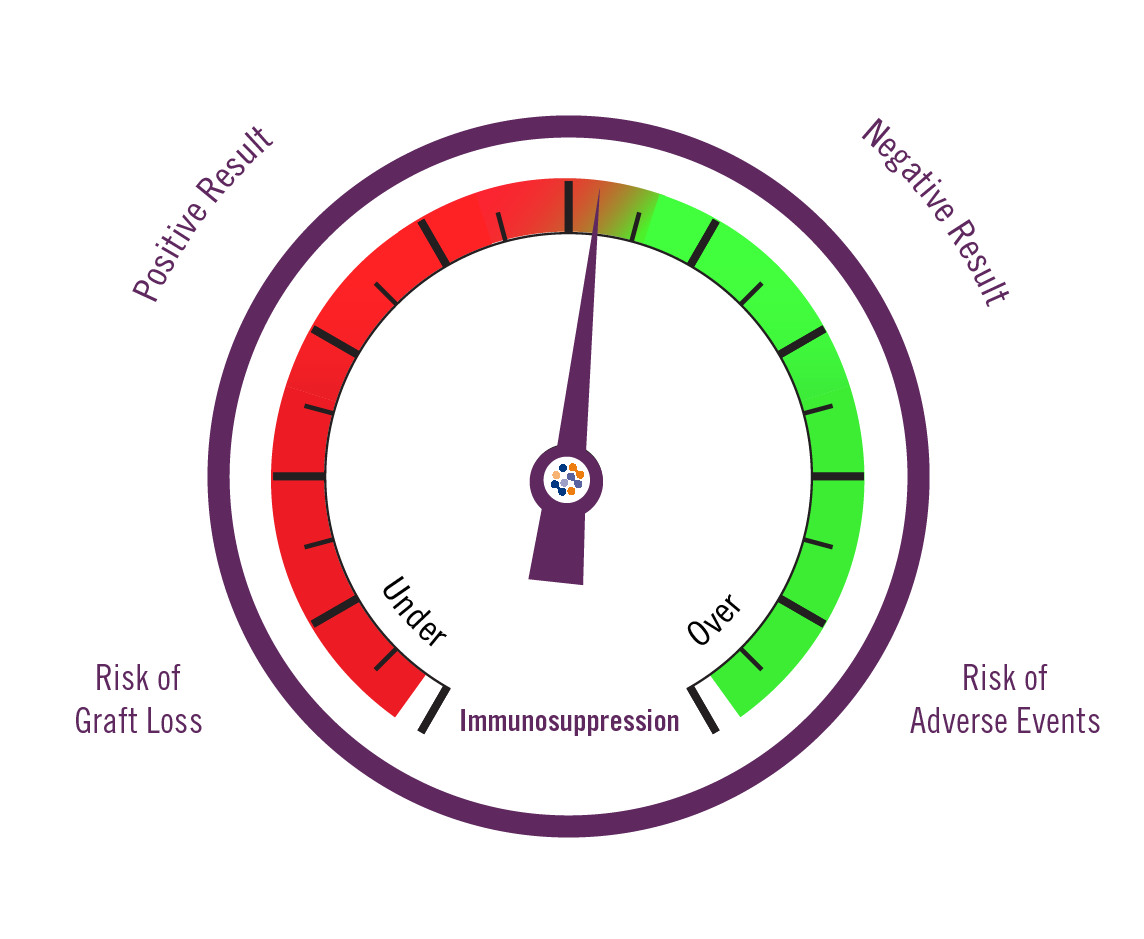
Biomarkers provide critical information that help guide clinical decision making and personalize patient care.

83% NPV
Negative Predictive Value
43% PPV
Positive Predictive Value
References:
- Tasdogan BE, et al. EJOHG 2019; 9 (2): 96–101.
- Kwong AJ et al. Am J Transpl 2022; 22 (S2): 204–309.